
| Home | Syllabus | Schedule | Lecture Notes | Extras | Glossary |

| Home | Syllabus | Schedule | Lecture Notes | Extras | Glossary |
We began by reminding all students to stay on top of this course, checking the schedule to make sure that they know when reading quizzes and homework assignments are due. Most students are reading the lecture notes.
The deadline for a short essay (no more than 1,000 words) on what you might learn from having your genome analyzed is midnight on Friday, September 13. The best assay will win a free genome analysis from 23andMe. This is not a class assignment, and no extra credit will be awarded for participation. You will not be required to share any of the results of the analysis of your genome with anyone, ever. You can read about one person's experience at OpenHumanGenome.
So far, on our concept map, we know from our introduction to Mendelism that genes are hereditary units that are not affected by their passage through hybrids. We know that during cell division (either meiosis or mitosis), cells go to a lot of trouble to distribute chromosomes to daughter cells. We suspect, from observing the effects of extra chromosomes on human development in aneuploid syndromes, that genes are on chromosomes. We will take up this problem in the next lecture. We also know that genes are made of DNA and generally encode proteins.
We reviewed that Mendel's key insight was in his selection of research materials: discrete traits that are very different between two parental lines, for which hybrids entirely resemble one parent. The genes for these traits do not interact with other genes or with the environment. The theory that Mendel developed from his experiments is a theory concerned with how genes are transmitted to offspring, not how genes influence traits. It follows that if we discover variants that don't act like Mendel's variants, his theory is not challenged unless these traits are transmitted by a mechanism beyond his discoveries.
It is useful at this point to consider a concept map for traits, shown below.
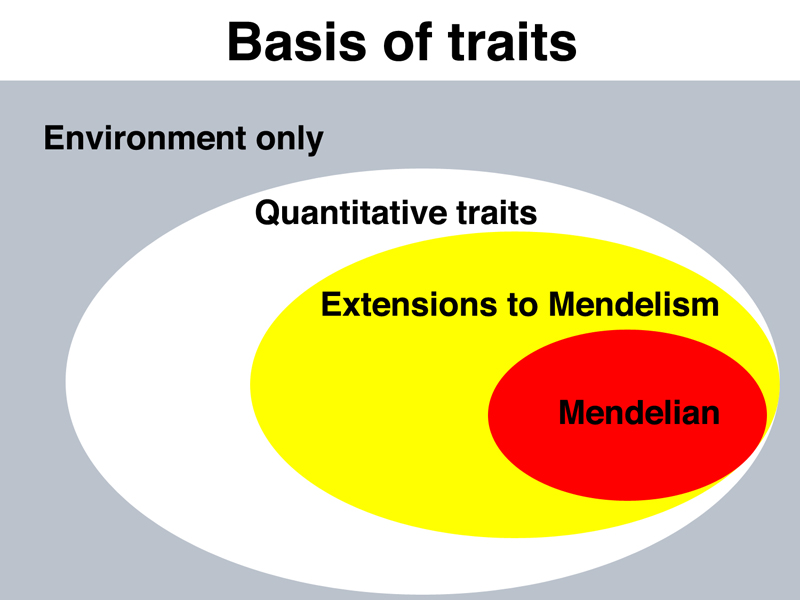
Some traits are completely determined by our environment, for example what language we speak. We have briefly covered quantitative traits in prior lectures. We know that quantitative traits like height, weight, blood pressure, and so on, exhibit a continuous range of variation, result from an interaction of genes and the environment, and are generally controlled by a large number of genetic factors, each contributing slightly to the overall phenotype.
At the other end of the spectrum are Mendelian traits, single genes that have a major impact on phenotype, that are not much affected by the environment or by other genes. Today's lecture deals with extensions to Mendelism, considering genes that behave in ways beyond what Mendel observed. These genes are still inherited by the rules that Mendel described.
In thinking about the nature of variants that Mendel investigated, we can see that all of his traits were controlled by single genes. We saw some evidence last time from our discussion of waltzing mice that it is possible for a single gene to affect a number of traits. Waltzing mice, for example, exhibit waltzing behavior, are deaf, and have anatomical abnormalities in their inner ear. We can understand all of these aspects of the phenotype of waltzing mice by seeing that they are related. We also saw that there is another allele of this gene that causes mice to be deaf, but not to exhibit waltzing behavior. In the related syndrome in humans, Usher Syndrome, we saw common traits of deafness, vestibular dysfunction, and retinitis pigmentosa. We learned that variants of quite a large number of genes can cause Usher Syndrome, and that individuals with variants in one gene cannot be easily distinguished from individuals with variants in another gene by phenotype alone.
This introduces the subject of pleiotropy, in which single allelic variants produce a large number of distinct traits. While at first this seems to be beyond what Mendel observed, often when we are able to understand the underlying defect at the level of the protein affected, the different traits can be seen to be related.
There is no better example of pleiotropy than Marfan Syndrome, a dominant disorder that is characterized by departure from a normal phenotype with respect to a large number of apparently disparate traits:
While several students in the class had heard of Marfan Syndrome, those who had not could not immediately imagine a mechanism that united all of these defects. Marfan Syndrome seems at first to be a puzzling example of pleiotropy.
In the early 1990s, the gene underlying Marfan Syndrome was identified as fibrillin (FBN1). Fibrillin is a protein component of the extracellular matrix in connective tissue. Both elastic fibers and collagen fibers contain crosslinked arrays of hundreds of molecules of fibrillin, which is secreted from cells and assembled in the extracellular matrix. Once we know this, it is easy to imagine how variants of the fibrillin protein might act as dominant alleles. A fibrillin protein that has an altered sequence and structure might ruin the properties of a large macromolecular assembly that contained the normal protein.
All of the various aspects of the phenotype of Marfan Syndrome are united by the understanding that connective tissue is defective in Marfan Syndrome. Long bones grow during development guided by connective tissue. The overgrowth of long bones explains the long arms, long fingers, sunken chest (overgrowth of ribs), and kyphoscoliosis. The lens of the eye is held in place by connective tissue, and failure here causes ectopia lentis. The formation of dissecting aortic aneurysms is also explained by defective connective tissue.
A student asked about the differences in phenotype between different individuals with Marfan Syndrome. As we were about to discuss multiple alleles in connection with cystic fibrosis, I answered briefly that there were a number of different alleles of FBN1 that cause Marfan Syndrome, and that they differed with respect to their effects on phenotype. In addition, it is possible that the genetic background of an individual (all the other genes present in the genome) affects the phenotype for an allele, and there may also be interactions with the environment.
You can read more about Marfan Syndrome at OMIM.
Another example of pleiotropy is taillessness in Manx cats. Manx cats have short tails or no tails at all, but it is not possible to develop a line of cats in which taillessness breeds true. Two tailless Manx cats will always produce some tailed offspring when mated. If the offspring with tails are mated, there is no further production of tailless offspring from such pairs. This suggests that taillessness among Manx cats is a dominant trait, which is confirmed when a tailless Manx is crossed to a long-tailed cat in a testcross: half of the offspring are tailless. Yet for some reason, even though there should be 1/3 TT and 2/3 Tt among the tailless offspring of a mating of tailless Manx cats, a true-breeding line cannot be established.
This is because TT cats die in utero. The T allele that confers dominant taillessness is itself a recessive lethal.
This example points out that it is wrong to ask whether an allele is dominant or recessive without selecting a particular other allele for comparison, and without specifying the trait in question. With respect to taillessness, T is dominant to t. With respect to lethality, T is recessive to t.
Penetrance is an expression of the fraction of individuals of a given genotype that show a particular phenotype. An allele that is fully penetrant shows 100% of individuals showing at least some aspect of the phenotype. An allele that is less than fully penetrant shows at least some individuals whose genotype we would infer incorrectly based on their phenotype.
Expressivity is a measure of the extent of phenotypic variation among individuals with the same genotype. We saw the example of piebald white spotting in dogs, where some dogs have only white socks, some have extensive white fur, while others are almost fully white, despite all having the same genotype.
We looked at an example of incomplete penetrance in a pedigree, where an individual judged to be of normal phenotype transmitted a rare dominant disorder that affected one of her parents to multiple offspring.
Finally, we looked at the hands of an individual with preaxial polydactyly. One hand had a partially duplicated thumb, while the other hand was normal. The class agreed that this was clearly an example of variable expressivity, because the individual's hands were discordant. It was not clear from this one photo whether this dominant trait shows incomplete penetrance as well, with individuals who are obligate carriers showing no phenotype.
We already know from our discussion of waltzing mice that it is possible to have more than two alleles for a single gene present in a population, even though any individual never carries more than two alleles. We showed pictures of Siamese cats and Himalyan mice, which are white with color at the "points," the nose and part of the face, the ears, the feet and tail. This pattern of pigmentation can be seen in mice, rats, cats, and rabbits, as these highly unusual variants were prized among domesticated mammals. The phenotype is due to an allele of the tyrosinase gene, the same gene responsible for albinism. The Siamese or Himalayan variant is allelic to albino. The tyrosinase protein encoded by these variants is sensitive to temperature, and does not function at the full body temperature of the animal. Only the cooler parts of the body (the "points") are pigmented.
We can compare the phenotypes of all possible combinations of the three alleles. Let's call them C (full color), cs (Siamese), and c (albino or colorless). The phenotypes of all possible genotypes are shown below.
| Genotype(s) | Phenotype |
| CC, Ccs, Cc | black |
| cscs, csc | Siamese |
| cc | albino |
Once again, it only makes sense to describe an allele as dominant or recessive based on a comparison to another specific allele. The full-colored C allele is dominant with respect to both cs and c. The Siamese allele cs is recessive with respect to C and dominant with respect to c. The albino allele c is recessive to both C and cs.
An extreme example of mulitple alleles is the gene responsible for cystic fibrosis, CFTR. Cystic fibrosis is well known as the most common genetic disorder leading to early death among people of European descent. In some populations, about one in 15 people is a carrier for a recessive variant that causes cystic fibrosis. Apparently, the recessive variants confer a selective advantage upon heterozygotes for some reason.
People who are homozygous or compound heterozygous for variant alleles of the gene causing cystic fibrosis exhibit a wide array of symptoms. The worst feature is the accumulation of thick mucus in the lungs. The mucus provides an ideal environment for the growth of bacteria, and cystic fibrosis patients are hospitalized frequently because of lung infections. They have blocked pancreatic ducts. Because the pancreas provides enzymes needed to digest food, patients with cystic fibrosis have special nutritional needs. Males with cystic fibrosis often have congenital bilateral absence of the vas deferens and are sterile. People with cystic fibrosis also have excess salt in their sweat. Like Marfan Syndrome, this collection of disparate traits seems at first to be a perplexing example of pleiotropy, but it is easier to understand once we know the nature of the responsible gene.
The gene underlying cystic fibrosis is CFTR, which encodes a chloride channel. Charged ions cannot pass through a lipid bilayer unless there is a protein pore. The CFTR gene encodes a pore that allows regulated passage of chloride and bicarbonate ions through plasma membranes.
There are over 1500 variant alleles of the CFTR gene known. I selected a few of them to show that different alleles manifest different aspects of the phenotype of cystic fibrosis. The table below shows a few different traits associated with the phenotype of cystic fibrosis, indicating for each variant the phenotype produced. The variants are named using an allele designation that indicates the position of the amino acid in the protein, the amino acid considered to be wild type at that position, and the new amino acid at that position (X indicates chain termination).
| Allele | Lung | Pancreas | Salty Sweat | Vas deferens |
| CFTR-F508del | - | - | - | - |
| CFTR-S1455X | + | + | - | + |
| CFTR-R75Q | + | +/- | + | + |
| CFTR-D1270N | + | + | + | - |
| CFTR-M470V | + | + | + | + |
CFTR-F508del. This is the most common variant allele of CFTR and appears to result in a complete loss of protein function. A single amino acid, phenylalanine at position 508, is deleted. Patients homozygous for this variant, or compound heterozygotes who carry this variant and one of the many other loss-of-function alleles manifest full-blown cystic fibrosis, with symptoms affecting their lungs and pancreas. They have salty sweat and males are sterile.
CFTR-S1455X. This allele is unusual in that patients homozygous for this allele, or compound heterozygotes for this allele and a loss-of-function allele like CFTR-F508del, have salty sweat but no other aspect of the phenotype of cystic fibrosis.
CFTR-R75Q. This allele, in the presence of a variant allele of another gene, predisposes to pancreatitis. This allele affects bicarbonate transport but not chloride transport. There are no other symptoms of cystic fibrosis. I am heterozygous for this allele (I'm fine, it's recessive).
CFTR-D1270N. This allele only causes congenital absence of the vas deferens in males; there are no other aspects of the phenotype of cystic fibrosis.
CFTR-M470V. This is a "silent" variant, with an allele frequency of almost 50% in some populations. It has no effect on the phenotype. This kind of allele would never have been detected by searching for phenotypic variation; discovery of silent alleles is commonplace with widespread genomic sequencing of individuals.
The point of this very selective presentation of CFTR alleles is to show that different alleles of CFTR can produce different phenotypes. Unlike the three alleles of waltzer (wild type, waltzer, and adult hearing loss) or the three alleles of tyrosinase (wild type, Siamese, and albino), we cannot order these alleles into a series with increasingly severe phenotypes. Also, the CFTR-M470V allele shows that calling a single allele the wild-type allele is an oversimplification.
You can read more about cystic fibrosis and CFTR at OMIM.
Mendel's traits were affected by two different alleles of seven genes. In each case, the hybrid is entirely like one parent; one allele is fully recessive and the other is fully dominant. This was a deliberate choice on Mendel's part that makes the experiments easier to interpret.
In four o'clocks (Mirabilis), there is a variant with white flowers as opposed to the normal purple flowers. When white and purple are crossed, the hybrids have pink flowers, halfway between the two parents in color. While at first this sort of observation looks like evidence for blending inheritance, when the F1 flowers are selfed, there is a 1:2:1 ratio of purple:pink:white in the F2. This is a beautiful demonstration of the First Law, the principle of segregation. If Mendel had found a trait like this in peas, he could have saved himself the trouble of testcrosses to show that the 3:1 F2 ratio was really a 1:2:1 ratio. This kind of trait, where the hybrid is partway between the two parents with respect to its phenotype, is called incomplete dominance or semidominance.
The human ABO blood type is controlled by a series of three classes of alleles of a single gene, as shown in the table below.
| Genotype | Phenotype |
| IAIA, IAi | A |
| IBIB, IBi | B |
| IAIB | AB |
| ii | O |
These alleles control the exact chemical nature of a carbohydrate that is attached to a protein found on the surface of red blood cells. A particular carbohydrate, one monosaccharide unit shorter than A or B, is the O antigen, which is really the absence of the A antigen and the B antigen. If a particular monosaccharide is added, this becomes the A antigen; if a different one is added, it's the B antigen.
The i allele behaves as a recessive. The A and B alleles are both dominant to i. With respect to each other, they are codominant: both A and B antigens are present on the surface of red blood cells from IAIB individuals, who have blood type AB.
Once we had established the rules for inheritance of ABO blood type, we played a quick round of Job Interview @ Who's Your Daddy? In this game, I showed a pedigree with blood types, and asked whether the first individual indicated could have been the father of the second individual indicated. The game illustrates that in science, we can only invalidate, but not prove, a hypothesis. In some of the pedigrees, we can say that the individual in question could not be the father of the indicated child. In the other cases, while paternity is not ruled out, it is not established. Paternity could be definitively established using additional DNA markers, which we will cover later in the course.
Before we began the game, I stated that the company had two kinds of positions open. They needed people to drive the sample trucks around various neighborhoods looking for business. For the right kind of person, this represents a great opportunity to have a job where they would meet interesting people, who might not be at their best at the moment. The company also needed people to work in the air-conditioned comfort of the DNA sequencing labs, where robots do most of the actual work and there is an awesome break room with broadband internet access. The pay for the lab job is also great. The company uses the game to sort job applicants.
All pedigrees from the game are shown below, with answers indicated.
| Job Interview @ Who's Your Daddy? | |
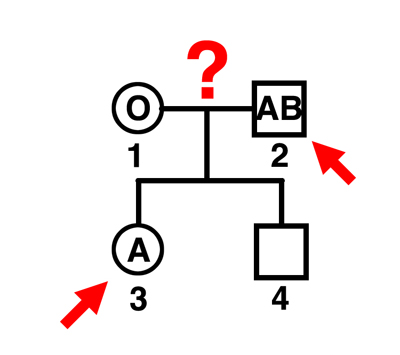 |
Q. Can individual 2 be the father of individual 3?
A. Yes. The mother can only contribute an O gamete. This man can father A or B children by this mother, and the indicated child is A. |
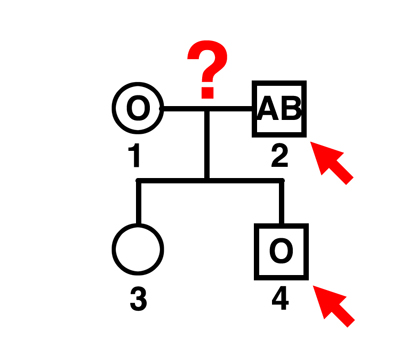 |
Q. Can individual 2 be the father of individual 4?
A. No. The mother can only contribute an O gamete. This man can father A or B children by this mother, and the indicated child is O. |
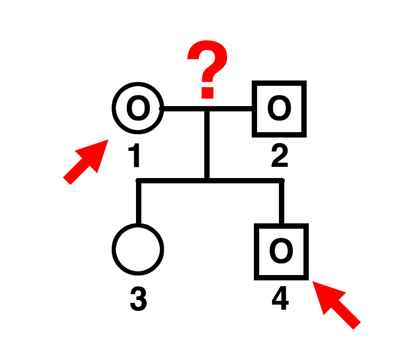 |
Q. Can individual 1 be the father of individual 4?
A. No. Girls can't be fathers. |
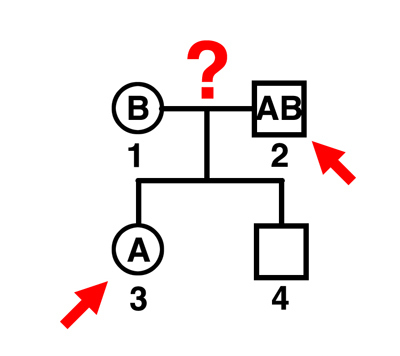
 |
Q. Can individual 2 be the father of individual 3?
A. No. The mother can only contribute an O gamete. This man can father only O children by this mother. |
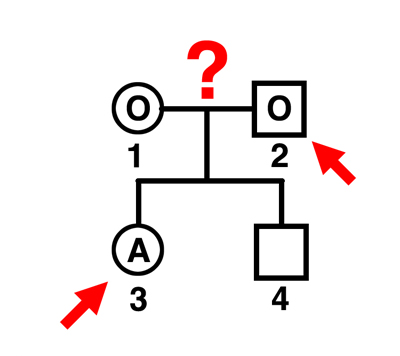
 |
Q. Can individual 2 be the father of individual 4?
A. Yes. The mother can only contribute an O gamete. This man can father only O children by this mother. |
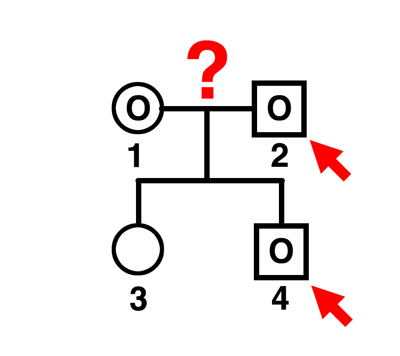
 |
Q. Can individual 2 be the father of individual 3?
A. Yes. The mother can contribute a B gamete (if she is BB) or possibly an O gamete (if she is BO). This man can father A (AO), B (BB), or AB children by this mother. |
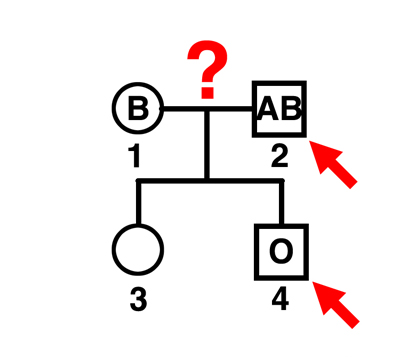 |
Q. Can individual 2 be the father of individual 4?
A. No. The mother can contribute a B gamete (if she is BB or BO) or possibly an O gamete (if she is BO). This man can father A (AO), B (BB), or AB children by this mother. |
From the overall performance of the class in this game, it looks like we will fill all the lab jobs right away, and will have to look harder for friendly, outgoing people to drive the truck.
Mendel's traits all affected different traits. It is interesting to see what happens when we have more than one gene affecting the same trait, in this case coat color in the mouse. We have already used black (CC) and albino (cc) mice in our initial discussion of Mendelism. Here we will use the old mouse symbol C instead of the A that we first used, or the current nomenclature, Tyr and Tyrc. There are two other genes known to mouse fanciers that affect coat color: B (black) vs. b (brown), and D (non-dilute) vs. d (dilute). Brown mice (bbCCDD) are a nice warm brown color, dilute mice (BBCCdd) are grey, and brown dilute mice (bbCCdd) are a color described as lilac or champagne by mouse fanciers, but we will just call it brown dilute.
The picture below shows various mouse genotypes and the resulting coat color.
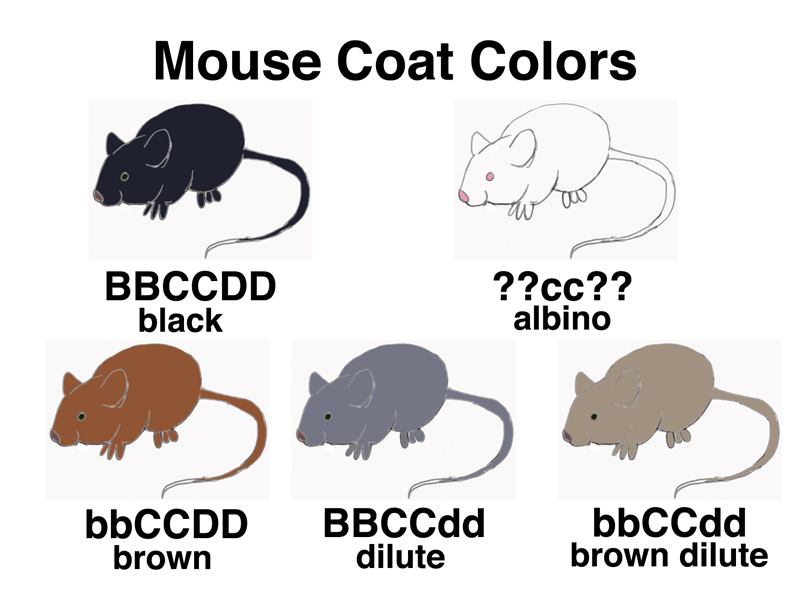
There are two examples of gene interaction among these five types of mice. The first is the interaction of brown and dilute. When you look at a brown dilute mouse, you see that it is a combination of dilution of pigment that turns black to gray, and the browning of pigment that truns black to brown.
The second type of gene interaction will require testcrosses to explore further: the interaction of albino with any other coat color gene. Notice that we have indicated the genotype of the albino mouse as ??cc??. We know that it is homozygous for the recessive albino allele (cc), because it is an albino. The only pigment in mouse fur is melanin. When melanin synthesis is blocked by a loss-of-function allele of the tyrosinase gene, we can't tell what color the mouse would have been if it were able to synthesize melanin. Mice that are BBccDD, bbccDD, BBccdd, and bbccdd all look exactly the same: they are albinos.
How do we describe the relationship of the albino allele of tyrosinase to the other coat color genes B and D? Several students wanted to say that albino is dominant to brown or dilute, but this is not correct. The terms dominant and recessive (and semidominant and codominant) only apply to alleles of the same gene. Albino (c) is recessive to colored (C), but we can't use this term to describe the relationship of albino (c) to brown (b) or dilute (d), because those are different genes.
The term that applies in this case is epistasis. Albino is epistatic to brown and dilute, meaning that the recessive albino allele masks the phenotype of mice with respect to brown and dilute. The opposite term is hypostasis. Brown and dilute are hypostatic to albino.
In order to determine the genotype of albino mice with respect to brown and dilute, we will conduct testcrosses. The ideal testcross strain should be homozygous for colored (CC) so that the hybrids cannot be albino. It should also be homozygous for the recessive alleles for brown (bb) and dilute (dd). The outcomes of testcrosses of four different albino inbred strains to bbCCdd are shown below.
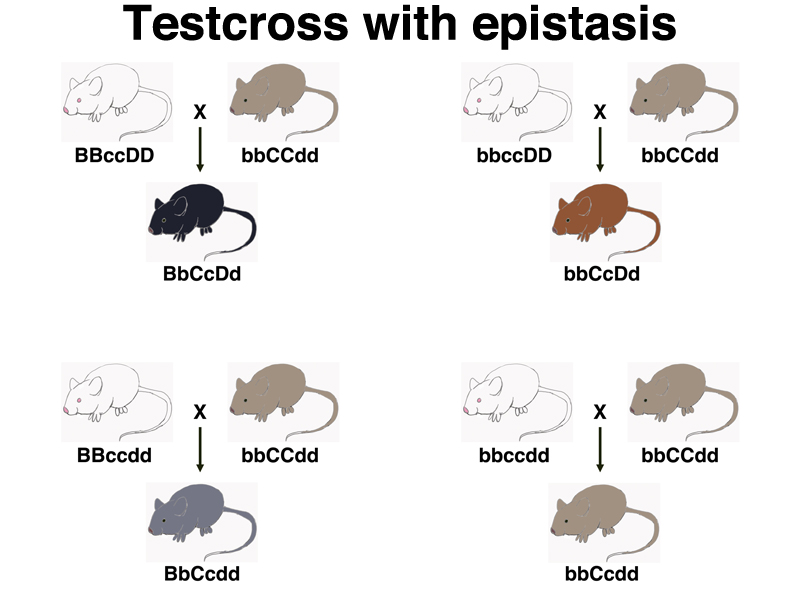
Note in the drawing above that the genotype of the albino is only apparent to us once we see what kind of progeny are produced, although the drawing is labeled. In these four cases, the albino strain is homozygous, because it is an inbred strain.
We can use the testcross to determine the genotype of any albino animal, as shown in the example below.

Students were able to infer that the genotype of the albino in this example was BbccDD. In addition, they were able to predict that the ratio of black to brown in this testcross would be 1:1. It is left as an exercise to the student to predict the outcomes of testcrosses of nonhomozygous albinos with different genotypes, such as BBccDd and BbccDd.